FROM EDITOR
CLINICAL PHARMACOGENETICS
What is already known on this topic?
Statins, particularly atorvastatin, are the cornerstone of lipid-lowering therapy for coronary heart disease (CHD).
There is significant interindividual variability in patient response to statin therapy.
Atorvastatin is primarily metabolized by the CYP3A4 enzyme in the liver.
Genetic polymorphisms in the CYP3A4 gene can alter enzyme activity and potentially affect drug efficacy and safety.
However, previous research on the impact of specific CYP3A4 variants, especially rs2740574 (A/G), has yielded contradictory results.
What does this article add?
This study provides the first data on the frequency of three CYP3A4 polymorphisms in a CHD patient cohort from Arkhangelsk, a northern region of Russia, revealing a significantly higher frequency of the G allele (8.3%) compared to the general Russian (4%) and European (3.63%) populations.
It demonstrates that carriers of the G allele (rs2740574) show a significantly more pronounced reduction in total cholesterol and LDL-C after 2-4 months of atorvastatin therapy.
It confirms that two other polymorphisms (Leu293Pro and Phe189Ser) are extremely rare in this specific population, limiting their clinical utility.
How might this influence clinical practice in the foreseeable future?
The findings support the potential for implementing pharmacogenetic testing to personalize statin therapy.
Identifying patients with the G allele could help clinicians predict a stronger response to atorvastatin, potentially guiding the choice of statin or initial dosing strategy.
This could lead to more efficient achievement of target LDL-C levels, reducing the need for dose adjustments or combination therapies.
However, due to the study's limitations (small sample size, single-center design), larger confirmatory studies are needed before these findings can be widely adopted into routine clinical guidelines.
Relevance. Pharmacogenetics is one of the leading areas of personalized medicine, allowing the prediction of the effectiveness and safety of medicines in a particular patient. This study analyzed the presence of CYP3A4 gene allele mutations and their relationship with the effectiveness of therapy.
Objective. This study aimed to evaluate the effect of the genetic polymorphism A/G (rs2740574) of the CYP3A4 gene, as well as polymorphisms CYP3A4_2 Leu293Pro (rs28371759) and CYP3A4 Phe189Ser (rs4987161) in patients with coronary heart disease on the lipid-lowering efficacy of atorvastatin in real-world practice.
Materials and methods. This study included 96 patients with coronary artery disease who received atorvastatin therapy. Molecular genetic analysis of CYP3A4 gene polymorphisms was performed using real-time polymerase chain reaction. Statistical data processing was performed using STATA 14 software.
Results. The frequency of the G allele (rs2740574) in the studied sample was 8.3 %, which differed significantly from that of the all-Russian (4 %, p = 0.0095) and European (3.63 %, p = 0.0005) populations. The frequency of the minor C allele for the CYP3A4_2 Leu293Pro (rs28371759) polymorphism was 0.5%, which was significantly different from the global and European frequencies (p < 0.001). Polymorphism CYP3A4 Phe189Ser (rs4987161) was not detected in the sample. Carriers of the G allele (rs2740574) (n = 15) showed a significant decrease in total cholesterol (from 5.38 ± 1.49 to 3.23 ± 0.96 mmol/l, p = 0.0019) and LDL-C (from 3.54 ± 1.17 to 1.58 ± 0.62 mmol/l, p = 0.0004) during atorvastatin therapy. The effect of other polymorphisms on the lipid profile could not be assessed due to their low prevalence.
Conclusion. Unique frequencies of CYP3A4 gene alleles, which differ from the reference populations, have been identified in patients with coronary heart disease in Arkhangelsk. The presence of the G allele (rs2740574) is associated with a more pronounced lipid-lowering response to atorvastatin therapy. The findings highlight the importance of pharmacogenetic studies for the personalization of statin therapy.
What is already known about this topic?
Role of GST: Genes encoding glutathione S-transferases (GSTM1 and GSTT1) are involved in the detoxification of xenobiotics, including chemotherapeutic agents (e.g., cyclophosphamide, doxorubicin, taxanes) used in breast cancer (BC) treatment.
Polymorphism: The most studied variants are the "null" (deletion) polymorphisms of these genes, which result in a complete loss of enzyme activity.
Contradictory Data: Previously published findings have been inconsistent:
Some studies indicated that "null" genotypes improve chemotherapy efficacy (better response, reduced relapse risk) due to slower clearance of cytostatics.
Other research (including meta-analyses) found no significant association between these polymorphisms and patient survival.
Most previous studies lacked detailed stratification by biological tumor subtype and disease stage.
What is new in the article?
Stratification by Clinical Features: For the first time in a Russian population (Primorsky Krai), a detailed analysis of chemotherapy efficacy was conducted based on GSTT1 and GSTM1 deletion status, with patients stratified not only by age but also by disease stage and molecular-biological subtypes of BC.
Specific Associations:
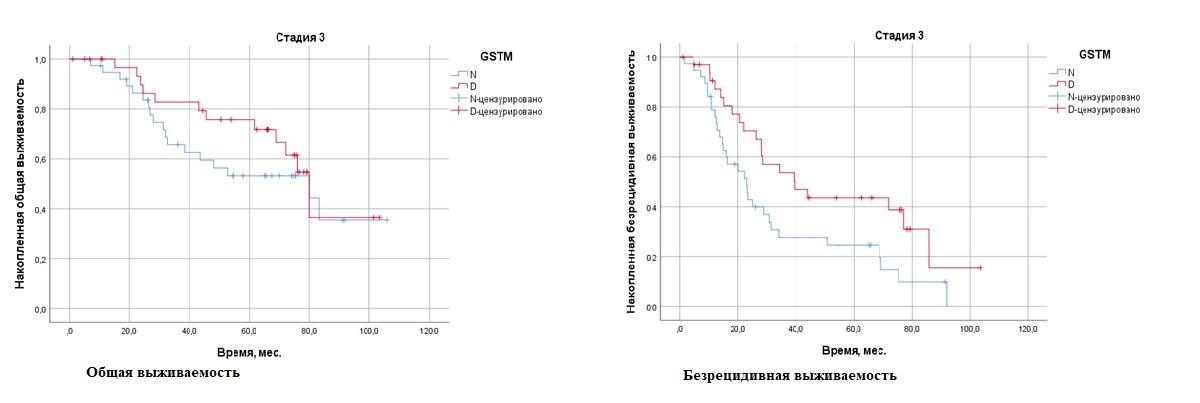
Stage III: The presence of "null" GSTM1 and GSTT1 genotypes was statistically significantly associated with improved relapse-free survival in patients with stage III disease. The risk of relapse was reduced by 0.52-fold and 0.4-fold, respectively.
Hormone-Dependent Subtypes:
In luminal A and luminal B HER2-negative subtypes, the GSTT1 deletion was associated with no lethal outcomes (significant increase in overall survival).
In the luminal B HER2-negative subtype, the GSTM1 deletion also significantly reduced the risk of relapse.
In the luminal B HER2-positive subtype, no deaths were recorded among patients with the GSTM1 deletion.
Age: No statistically significant effect of age at disease onset was found on the association between the polymorphisms and survival.
How can this affect clinical practice in the foreseeable future?
Refining Prognosis: Pharmacogenetic testing for "null" GSTT1/GSTM1 genotypes could become an additional tool for predicting chemotherapy efficacy, particularly in patients with locally advanced (stage III) and hormone-dependent (luminal subtypes) BC.
Personalizing Therapy: Clinicians could consider a patient's genetic status when choosing a treatment strategy:
Patients with "null" genotypes (slower drug metabolism) may derive a better antitumor effect from standard chemotherapy, providing greater confidence in administering intensive regimens.
This knowledge could help avoid unnecessary treatment escalation or, conversely, justify its use in high-risk patients (stage III) if they have a genotype favorable for treatment response.
Economic Feasibility: Evidence-based prescription of costly drugs or intensive regimens based on genetic test results (considering proven benefits for specific patient subgroups) could improve cost-effectiveness in healthcare.
Limitation: The authors note that the sample size for some subtypes was small, requiring confirmation in larger cohorts before these markers can be incorporated into routine clinical guidelines.
Relevance. The steady increase in the incidence of breast cancer (BC), as well as associated mortality and disability of the population, determines the relevance of the search for effective treatment and prevention of this pathology.
Objective. Evaluation of differences in the effectiveness of chemotherapy for BC depending on the age, stage, and biological subtype of the tumor, considering the deletion status of the GSTT1 and GSTM1 genotypes in patients.
Materials and methods. Data from 132 patients with BC who received chemotherapy treatment from 2013 to 2021 were analyzed. Polymorphic GSTM1 and GSTT1 variants were genotyped using multiplex polymerase chain reaction, followed by analysis of the melting curves of the reaction products.
Results. The presence of the "null" genotype of GSTM1 and GSTT1 reduced the risk of relapse in patients with stage III disease by 0.52 times (95 % CI 0.29–0.89, p = 0.023) and 0.4 times, respectively (95 % CI 0.098–0.99, p = 0.049). Patients with luminal B HER2-positive breast cancer and GSTM1-0 had no fatalities. The risk of relapse was reduced in women with luminal B HER2-negative breast cancer subtype in the GSTM1-0 genotype group. In patients with luminal A, the overall survival (OS) with the GSTT1 wild type was 75.5 (±12.3) %, with GSTM1-0 there were no lethal outcomes (OR = 0.034, 95 % CI 0.02–0.045, p = 0.001), with luminal B HER2-negative subtypes, OS with the GSTT1 wild type was 69.9 (±8.5) % versus no lethal cases with the null genotype (OR = 0.035, 95 % CI 0.025–0.044, p = 0.001)).
Conclusion. The results of our study showed a significant effect of the GSTT1 and GSTM1 gene deletion polymorphism on relapse-free survival in patients with stage III disease and hormone-dependent breast cancer.
What is already known about this topic?
Variability in morphine response: The pharmacokinetics and analgesic effect of morphine are characterized by high interindividual variability, making it difficult to select a safe and effective dose.
Role of the ABCB1 gene: The ABCB1 (MDR1) gene encodes the P-glycoprotein (P-gp) transporter, which limits the penetration of morphine across the blood-brain barrier and influences its absorption and elimination.
Influence of polymorphisms: Certain single nucleotide polymorphisms (SNPs) in the ABCB1 gene (rs1128503, rs2032582, rs1045642) can alter the expression or function of P-gp.
International data: Studies in other populations (e.g., Italian) have shown a link between these polymorphisms, especially rs1045642 (C3435T), and the effectiveness of morphine analgesia, as well as the risk of developing side effects.
What is new in the article?
Data from a Russian cohort: For the first time in a Russian population of palliative care patients (86 individuals), the study confirms the association between three ABCB1 polymorphisms and morphine plasma concentrations (not just with the clinical effect).
Quantitative assessment: It shows that carriage of minor alleles (especially the homozygous TT genotype for rs1045642 and rs2032582) is associated with statistically significant higher steady-state morphine concentrations in plasma.
Example: For rs1045642 in the 80–100 mg/day dose group, the median concentration in TT carriers was 151.8 nmol/L versus 83.7 nmol/L in CC carriers.
Dose-dependent effect: The study found that genotype-specific differences in concentration become most pronounced and reach statistical significance in patient groups receiving medium and high doses of morphine (40–60 mg and 80–100 mg/day).
How can this affect clinical practice in the foreseeable future?
Personalization of initial dose: Preliminary ABCB1 genotyping could help stratify patients into risk groups.
TT genotype carriers (rs1045642, rs2032582): May require lower starting doses and close monitoring due to the risk of drug accumulation and overdose.
CC genotype carriers ("wild type"): May demonstrate relative resistance to therapy, potentially requiring faster dose titration or consideration of alternative opioids.
Improved therapy safety: Accounting for genetic predictors could reduce the incidence of adverse events (drowsiness, respiratory depression, toxic effects) associated with high morphine concentrations in genetically predisposed patients.
Integration of testing into algorithms: The results support the feasibility of including pharmacogenetic testing for the ABCB1 gene in clinical guidelines and pain management algorithms in palliative oncology to optimize the efficacy/safety ratio.
Background. Morphine pharmacokinetics in cancer patients are characterized by high interindividual variability, partly due to genetic factors. The role of ABCB1 gene polymorphisms in modifying morphine exposure in this patient population has been poorly studied.
Objective. To evaluate the effect of ABCB1 gene polymorphisms rs1128503, rs2032582, and rs1045642 on steady-state plasma morphine concentrations in patients with cancer receiving palliative care.
Methods. This study included 86 patients with cancer treated in the Palliative Care Department of the Moscow Multidisciplinary Palliative Care Center of the Moscow Department of Health. All participants received stable oral morphine doses ranging from 30 to 100 mg/day. ABCB1 polymorphisms were genotyped using real-time polymerase chain reaction (PCR). Plasma morphine concentrations were determined using HPLC-MS/MS. Statistical analysis included an assessment of normality (Shapiro-Wilk test), nonparametric Mann–Whitney and Kruskal-Wallis tests, and χ², with p ≤ 0.05 considered statistically significant.
Results. In carriers of the TT genotype rs1045642, at a dose of 80–100 mg/day, the median morphine concentration was 151.8 nmol/L, exceeding the values in CT (110.4 nmol/L) and CC (83.7 nmol/L), with p = 0.097 (χ²), p ≤ 0.05 for pairwise comparisons. Similar trends were found for rs2032582 and rs1128503,
with significant differences between carriers of minor alleles. No adverse events related to the study intervention were reported.
Conclusion. Carriage of certain ABCB1 allelic variants is associated with increased exposure to morphine. Genetic predictors may facilitate individualized dosing in patients with cancer undergoing palliative care.
What is already known about this topic?
Patients undergoing total knee arthroplasty (TKA) have a high risk of venous thromboembolic complications (VTE), making prophylactic anticoagulation mandatory.
Direct oral anticoagulants (DOACs), including dabigatran, are effective for VTE prevention, but their efficacy and safety show significant interindividual variability.
The pharmacokinetics of dabigatran are known to be potentially influenced by genetic polymorphisms in genes responsible for its transport (ABCB1, encoding P-glycoprotein) and metabolism (CES1, encoding carboxylesterase).
What is new in the article?
Specific Genotype-Outcome Associations: The study provides clinical evidence linking specific genotypes to distinct outcomes:
ABCB1 3435TT genotype → Higher dabigatran concentration → Increased bleeding risk.
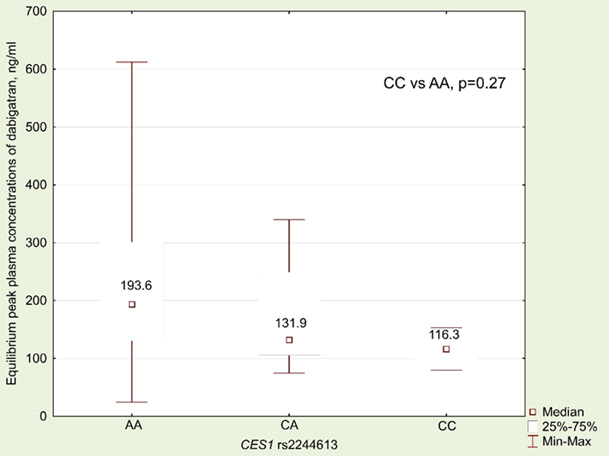
CES1 rs2244613 CC genotype → Lower dabigatran concentration → Increased thrombotic risk.
Identification of an "Unpredictable" Genotype: Heterozygous carriage of all three studied polymorphisms (ABCB1 rs1045642, ABCB1 rs4148738, and CES1 rs2244613) was identified as the most difficult profile for predicting either thrombosis or bleeding.
Identification of a "Favorable" Genotype: A specific combination (ABCB1 rs1045642 CC + ABCB1 rs4148738 TT + CES1 rs2244613 AA) was associated with high efficacy and safety, even in patients with a high baseline thrombosis risk.
Role of Drug-Drug Interactions: A case of severe bleeding was detailed, demonstrating how pharmacokinetic and pharmacodynamic interactions between dabigatran and commonly co-prescribed drugs (amlodipine, lisinopril, rosuvastatin, fondaparinux) can lead to serious adverse events.
Potential of the Thrombodynamics Test: The study suggests the thrombodynamics test is a promising tool for monitoring the real-world effectiveness of dabigatran therapy at an individual level.
How can this affect clinical practice in the foreseeable future?
Personalized Dosing: Preemptive genotyping for ABCB1 and CES1 could identify patients at high risk of bleeding (who might benefit from a lower dose) or those at risk of treatment failure (who might need a higher dose or a different drug).
Integration of Genetic Testing: Routine pharmacogenetic testing could become a standard step before initiating dabigatran for VTE prophylaxis in orthopedic surgery to predict individual response.
Increased Vigilance for Drug Interactions: The findings highlight the critical need to review a patient's full medication profile (especially statins and calcium channel blockers) to prevent dangerous interactions, particularly in genetically predisposed individuals.
A Multi-Pronged Personalized Approach: The study supports a future model of personalized anticoagulation that combines genetic testing, therapeutic drug monitoring (measuring drug concentrations), and functional coagulation assays (like thrombodynamics) to maximize both safety and efficacy.
Background. The effectiveness and safety of dabigatran for venous thromboembolism (VTE) prophylaxis show significant interindividual variability, partially attributed to pharmacogenetic factors.
Objective. To evaluate the influence of ABCB1 (rs1045642, rs4148738) and CES1 (rs2244613) gene polymorphisms on dabigatran pharmacokinetics and clinical outcomes in patients after orthopedic surgery.
Methods. The study included 60 patients who received dabigatran etexilate (220 mg/day) after total knee arthroplasty. Genotyping was performed by real-time PCR, and dabigatran plasma concentrations were measured by HPLC-MS/MS. Clinical outcomes (VTE, bleeding) were monitored.
Results. The ABCB1 3435TT genotype was associated with higher dabigatran concentrations and an increased bleeding risk, while the CES1 rs2244613 CC genotype correlated with lower concentrations and a higher thrombotic risk. A combination of ABCB1 (CC and TT) and CES1 (AA) genotypes demonstrated optimal efficacy and safety. Heterozygous carriage of all three polymorphisms had an unpredictable effect. A case of severe bleeding due to drug-drug interaction was described.
Conclusion. Genetic polymorphisms of ABCB1 and CES1 significantly influence dabigatran exposure and the risk of complications. Preemptive genotyping could personalize anticoagulant therapy to improve its safety and efficacy in post-operative patients.
ETHNIC ASPECTS OF PHARMACOGENETICS
What is already known about this topic?
Enzyme Role: Bedaquiline, a key drug for treating drug-resistant tuberculosis (DR-TB), is primarily metabolized in the liver by the cytochrome P450 isoenzyme CYP3A4.
Genetic Impact: Polymorphisms (genetic variations) in the CYP3A4 and CYP3A5 genes can alter enzyme activity. This can lead to variability in drug concentrations in the blood, affecting treatment efficacy and the risk of adverse effects.
Population Differences: The prevalence of these polymorphisms varies significantly between different ethnic groups (e.g., between Europeans and Asians). For instance, the CYP3A4*22 variant is rare in Asians but present in Europeans, while CYP3A5*3 is common worldwide but at different frequencies.
Knowledge Gap: Prior to this study, there were no data on the frequency of key polymorphisms (CYP3A4*22 and CYP3A5*3) among the indigenous population of Yakutia (Yakuts) and Russians living in that region who have TB.
What is new in the article?
First Data on Yakuts: This study provides the first-ever data on the distribution of CYP3A4*22 and CYP3A5*3 genotypes among Yakut patients with drug-resistant TB. It shows that Yakuts differ significantly from East Asian populations (e.g., Chinese, Japanese) in these genetic markers, despite their geographical proximity.
Unique Profile of Russians in Yakutia: Russians living in Yakutia have a CYP3A5*3 profile similar to the general European population. However, for the CYP3A4*22 gene, they show an almost complete absence of the "poor metabolizer" (T) allele, which distinguishes them from Europeans and aligns them more closely with Asian populations.
"Siberian" Genetic Signature: When comparing a combined "Siberian" group (Yakuts + Russians from Yakutia) to a general Eurasian group (Europeans + East Asians), the Siberians had:
A significantly lower frequency of the "poor metabolizer" allele for CYP3A4*22.
A significantly higher frequency of the "poor metabolizer" homozygous genotype (GG) for CYP3A5*3.
Predominant Phenotype: The study found that the vast majority of both Yakut and Russian patients are intermediate metabolizers for CYP3A-mediated drugs. The poor metabolizer phenotype is extremely rare (only 1% of Yakuts).
How can this affect clinical practice in the foreseeable future?
Personalizing Bedaquiline Therapy: The high frequency of the "poor metabolizer" CYP3A5*3 (GG) genotype in the Siberian population suggests these patients may have reduced bedaquiline clearance. This could lead to higher drug concentrations in their blood, potentially increasing both efficacy and the risk of toxicity.
Predicting Risks: Knowing a patient's genotype could help physicians identify those at higher risk for elevated bedaquiline concentrations and, consequently, a greater risk of adverse effects like QT interval prolongation on an ECG.
Justification for Genetic Testing: The significant interpopulation differences uncovered in this study strongly support the need for broader implementation of pharmacogenetic testing before prescribing bedaquiline, especially in regions with diverse and unique genetic backgrounds like Yakutia. This would enable a shift from standard dosing to truly individualized therapy, optimizing both treatment success and patient safety.
Background. Treatment of drug-resistant tuberculosis is associated with numerous medical and societal problems; therefore, the search continues for measures to improve treatment outcomes through the development of new drugs and chemotherapy regimens. Bedaquiline metabolized by the cytochrome P450 isoenzyme CYP3A4. CYP3A4 and CYP3A5 polymorphisms can lead to variability in bedaquiline plasma concentrations, which in turn affects treatment efficacy and safety. Currently, there are no data on the prevalence of CYP3A4*22 and CYP3A5*3 gene polymorphic variants among Yakuts and Russians with tuberculosis.
Objective. To study the carrier frequency of CYP3A4*22 (rs35599367) C>T and CYP3A5*3 (rs776746) A>G gene polymorphisms among Yakuts and Russians with drug-resistant tuberculosis compared to the normal variability of the studied gene allelic variants in East Asian and European populations.
Methods. A cross-sectional comprehensive study was conducted on 255 patients of Yakut and Russian ethnicity with drug-resistant pulmonary tuberculosis. CYP3A4*22 (rs35599367) C>T and CYP3A5*3 (rs776746) A>G polymorphisms were determined by real-time polymerase chain reaction. For comparative analysis, data on the carrier frequency of CYP3A4*22 and CYP3A5*3 allelic variants in East Asian and European populations were used.
Results. In patients with drug-resistant pulmonary tuberculosis, the frequency of the T allele of the CYP3A4*22 gene was significantly lower in the Siberian group compared to the frequency in the Eurasian group (p = 0.003). The wild-type genotype (CC) CYP3A4*22 was significantly more common in the Siberian group of patients than in the Eurasian group, whereas the heterozygous genotype (CT) was less represented in the Siberian population (p = 0.003). The AA genotype of CYP3A5*3 was less common in the Siberian group relative to its frequency in the Eurasian group (p = 0.021), the heterozygous AG genotype was much less frequently detected in the Siberian population of patients (p < 0.001), and the GG genotype predominated in the Siberian group and was less frequently determined in the Eurasian population (p < 0.001). The frequencies of alleles and genotypes of the CYP3A5*3 gene in patients with drug-resistant pulmonary tuberculosis did not differ between the Yakut and Russian populations. The distribution of CYP3A5*3 alleles and genotypes in the Russian population was the same as in the general European profile (p > 0.05). Differences were found between Yakuts and East Asians for all analyzed parameters (p < 0.05).
Conclusion. Features of the distribution of CYP3A4*22 and CYP3A5*3 gene allelic variants affect the rate of drug metabolism among patients of Yakut and Russian ethnicity. Differences in allelic variants and genotypes of CYP3A4 and CYP3A5 in Russian and Yakut populations with drug-resistant pulmonary tuberculosis can significantly modify clinical efficacy and the development of adverse reactions during bedaquiline treatment, as the CYP3A isoenzyme of cytochrome P450 plays a primary role in its oxidation.
ISSN 2686-8849 (Online)